The Pfizer Covid vaccine is already being administered to the public in the UK and the first doses have been given in the US ahead of a mass vaccination campaign on a global scale.
It is important to recognize that the Pfizer Covid vaccine has not been approved by the FDA. It has only received Emergency Use Authorization (EUA) meaning the vaccine has not gone through the standard process to get official approval from the regulatory agency.
Now Moderna’s experimental Covid vaccine is set to get the same Emergency Use Authorization allowing the shot to be distributed to millions of people.
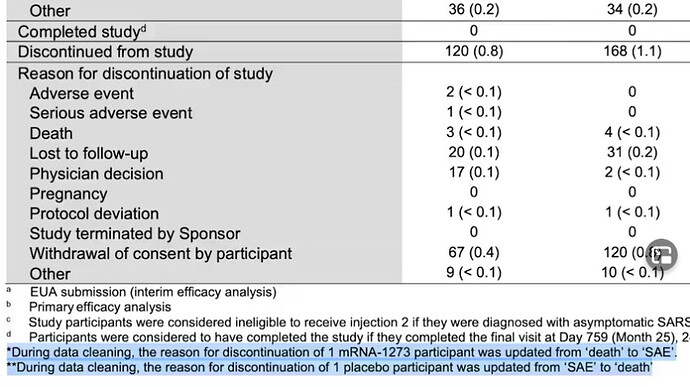
We have already witnessed short term adverse events (side effects) from the Pfizer vaccine. Truth is, nobody knows what the long term effects could be and it appears the public is being subjected to an experiment on a global scale.
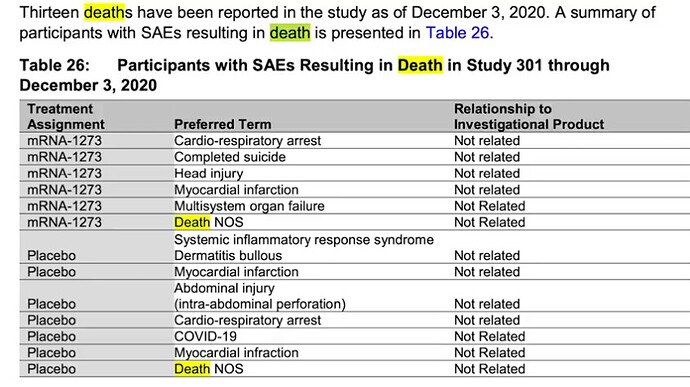
In this report, we examine discrepancies in the FDA Moderna report that was voted on by an advisory panel. The panel voted 20-0 recommending EUA.
Some of the discrepancies include cherry picked trial participants to achieve the desired results to gain EUA. As well as 13 total deaths in the trials, 6 in the vaccinated group and 7 in the placebo. Something the media refuses to address.
Page 6 has a graphic that says: unable to perform normal daily activities, unable to work, required care from doctor or health care professional
As of Dec 18, 3150 out of 112,807 are in this category
Morbidity and Mortality Weekly Report (MMWR)
On December 18, 2020, the Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for the Moderna COVID-19 (mRNA-1273) vaccine
Since June 2020, ACIP has convened 10 public meetings to review data on the epidemiology of COVID-19 and the potential use of COVID-19 vaccines, including the Moderna COVID-19 vaccine
…2 months of follow-up, indicate that the Moderna COVID-19 vaccine efficacy after 2 doses was 94.1% (95% confidence interval = 89.3%–96.8%) in preventing symptomatic, laboratory-confirmed COVID-19 among persons without evidence of previous SARS-CoV-2 infection, which was the primary study endpoint.
In other words people who dont have symptoms, (asymptomatic), after study they still dont have symptoms. In days gone by, this kind of people used to be called healthy or Not ill, therefor they didnt need toxic medicines.
Among vaccine recipients, reactogenicity symptoms, defined as solicited local injection site or systemic adverse reactions during the 7 days after vaccination, were frequent but mostly mild to moderate. Systemic adverse reactions were more commonly reported after the second dose than after the first dose and were more frequent and severe in persons aged 18–64 years than in those aged ≥65 years.
ACIP determined that COVID-19 is a major public health problem and that use of the Moderna COVID-19 vaccine is a reasonable and efficient allocation of resources. Whereas there might be uncertainty about how all populations value the vaccine, it was determined that for most populations, the desirable effects outweigh the undesirable effects, making the vaccine acceptable to implementation stakeholders.
Data on COVID-19–associated hospitalizations and deaths are limited at this time; however, a vaccine that effectively prevents symptomatic infection is expected to also prevent associated hospitalizations and deaths.
A lower risk of symptomatic COVID-19 was observed with vaccination compared to placebo (relative risk [RR] 0.06, 95% confidence interval [CI]: 0.03, 0.11, evidence type 1), corresponding to a vaccine efficacy of 94.1% (95% CI: 89.3%, 96.8%). This was observed with a median follow-up of two months, prompting concern for indirectness due to the short duration of follow-up (i.e., observed outcome of vaccine efficacy at two months does not directly inform vaccine efficacy for any duration longer than two months). The Phase III randomized controlled trial that generated this estimate is ongoing, thus the efficacy estimate may change with additional follow-up. However, given the strength of the association (i.e., high estimated vaccine efficacy), it is unlikely that the efficacy estimate for prevention of symptomatic COVID-19 would change substantially enough in the months following vaccination to fall below the FDA-defined efficacy threshold for an Emergency Use Authorization.
A list of names available as to who voted to vaccinate you listed in article above.
The Phase III randomized controlled trial accrued the number of cases needed to meet the primary endpoint of vaccine efficacy against symptomatic COVID-19 and the secondary endpoint of vaccine efficacy against severe COVID-19, based on a protocol definition that did not require hospitalization. Hospitalizations due to COVID-19 and deaths are less common, thus, Phase III trials may not be designed or statistically powered to evaluate differences between vaccine and placebo arms for these outcomes. However, for hospitalization due to COVID-19, a statistical difference was observed based on only 10 cases in the Phase III trial. Since robust direct evidence is not expected from early results from Phase III studies, vaccine efficacy in preventing hospitalizations due to COVID-19 and deaths may also be inferred from observed efficacy against symptomatic COVID-19.
Grade ≥3, or severe, local or systemic reactions 7 days following either vaccination, were reported by 22% of vaccine recipients, and occurred more frequently in the vaccine
Unsolicited Adverse Events
Reports of lymphadenopathy were imbalanced with 58 more cases in the vaccine group (64) than the placebo group (6); lymphadenopathy is plausibly related to the vaccine. Lymphadenopathy occurred in the arm and neck region and was reported within 2 to 4 days after vaccination. The average duration of lymphadenopathy was approximately 10 days. Bell’s palsy was reported by four vaccine recipients and none of the placebo recipients. The observed frequency of reported Bell’s palsy in the vaccine group is consistent with the background rate in the general population, and there is no basis upon which to conclude a causal relationship.
Serious Adverse Events
Serious adverse events were defined as any untoward medical occurrence that resulted in death, was life-threatening, required inpatient hospitalization or prolongation of existing hospitalization, or resulted in persistent disability/incapacity. The proportions of participants who reported at least 1 serious adverse event were 0.6% in the vaccine group and 0.5% in the placebo group. The most common serious adverse events in the vaccine group which were numerically higher than in the placebo group were appendicitis (7 in vaccine vs 2 in placebo), acute myocardial infarction (3 vs 0), and cerebrovascular accident (3 vs 1). Cardiovascular serious adverse events were balanced between vaccine and placebo groups. Two serious adverse events were considered by U.S. Food and Drug Administration (FDA) as possibly related to vaccine: shoulder injury possibly related to vaccine administration or to the vaccine itself, and lymphadenopathy involving the axilla contralateral to the vaccine injection site. Otherwise, occurrence of severe adverse events involving system organ classes and specific preferred terms were balanced between vaccine and placebo groups.
FDA Investigates Allergic Reactions to Pfizer COVID Vaccine After More Healthcare Workers Hospitalized
The FDA is investigating allergic reactions in “multiple states.” The agency acknowledged the reactions may have been caused by PEG, a compound in the Pfizer vaccine that CHD previously told the FDA could put millions of people at risk.
FDA’s Marks on Friday also acknowledged that allergic reactions to PEG could be “somewhat more common than previously understood.”
“At the very least, everyone should be screened for anti-PEG antibodies before getting the Pfizer and Moderna vaccines,” Kennedy said. “It is unconscionable that instead, the FDA and CDC are encouraging people to go ahead and risk a life-threatening anaphylactic reaction and just assume that someone will be on hand to save them.”
“If you have ever had a severe allergic reaction to any ingredient in a COVID-19 vaccine, CDC recommends that you should not get that specific vaccine. If you have had a severe allergic reaction to other vaccines or injectable therapies, you should ask your doctor if you should get a COVID-19 vaccine. Your doctor will help you decide if it is safe for you to get vaccinated.”
These guidelines are inadequate, Redwood said. “Many people who have never previously experienced any type of allergic reaction may be at risk of a severe reaction to the Pfizer and Moderna vaccines,” Redwood said. “That’s because millions of people may have developed anti-PEG antibodies as a result of having unknowingly been exposed to PEG in the past.”
According to research published in 2018, there are more than 1,000 products on the market — including pharmaceuticals, processed foods, cosmetics and disinfectants — that contain PEG. Peg is also the main ingredient in antifreeze.
“Anyone who’s had a colonoscopy, depending on what was used for preparation for the procedure, may have anti-PEG antibodies,” Redwood said, because many of those products contain PEG.
“It’s a simple blood test,” Redwood said. “Everyone considering getting the Pfizer or Moderna COVID vaccine should ask their doctor for a blood test to rule out anti-PEG antibodies before getting the vaccine.”
According to news reports, documents published by Pfizer and Moderna showed that people with a history of severe allergic reactions were excluded from the clinical trials, which explains why the adverse reaction to PEG didn’t show up in the trials.
From: https://zerohedge.whotrades.com/
Johns Hopkins Newspaper Removes Study Examining COVID Death Rate 2 hours ago
2 images 1 video
Johns Hopkins Newspaper Removes Study Examining COVID Death Rate
Authored by Benjamin Zeisloft via Campus Reform,
Johns Hopkins University’s student newspaper, the News-Letter, reported on a university stating that COVID-19 “had no effect on the percentage of deaths of older people” and that the virus “has also not increased the total number of deaths” in comparison to historical data . However, the paper later removed the article, stating that it had been used to support “dangerous inaccuracies” on social media.

Covid-19 Deaths- A Look at U.S. Data Nov 13, 2020
Assistant Director for the university’s Applied Economics program Genevieve Briand critically analyzed the net effect of COVID-19 on deaths in the United States based on historical data. Using information from the Center for Disease Control and Prevention, Briand identified the percentages of total deaths per age category both before and after the pandemic began.
“Surprisingly, the deaths of older people stayed the same before and after COVID-19,” said the News-Letter’s article.
“Since COVID-19 mainly affects the elderly, experts expected an increase in the percentage of deaths in older age groups. However, this increase is not seen from the CDC data. In fact, the percentages of deaths among all age groups remain relatively the same.”
Though deaths in categories like respiratory illnesses and heart disease seasonally rise and fall together in the United States, Briand noticed a strange trend.
“Instead of the expected drastic increase across all causes, there was a significant decrease in deaths due to heart disease,” in addition to “all other causes.” Additionally, “the total decrease in deaths by other causes almost exactly equals the increase in deaths by COVID-19.”
“All of this points to no evidence that COVID-19 created any excess deaths. Total death numbers are not above normal death numbers. We found no evidence to the contrary,” Briand concluded in her presentation. She told the News-Letter that “a decreased number of heart attacks and all the other death causes doesn’t give us a choice but to point to some misclassification.”
The News-Letter removed the article in late November after staff discovered its coverage of the study was "used to support dangerous inaccuracies that minimize the impact of the pandemic.” The paper linked a PDF of the original article with a watermark stating “Retracted by the News-Letter” to a statement explaining the decision.
The News-Letter explained that it made the decision independently and encouraged readers to take the article “in context with the countless other data published by Hopkins, the World Health Organization and the Centers for Disease Control and Prevention (CDC).”
The staff of the News-Letter referred Campus Reform to an op-ed written December 3 by the paper’s editorial board, which said that “the article should not have been deleted in the first place.”**
“Instead of temporarily removing it from our website, the News-Letter should have immediately retracted and provided a detailed explanation of the inaccuracies in Briand’s research,” explained the editors.
“We did not intend to silence Briand; instead, we sought to put her claims in conversation with findings from Hopkins, the World Health Organization and the CDC.”
Briand told Campus Reform that the News-Letter’s decision to remove the article was its own, and pointed out that she explained "during the presentation where I found and downloaded the data from, so anyone can easily replicate my analysis.”
Thousands of people have been unable to work or perform daily activities, or have required care from a healthcare professional, after getting the new COVID-19 vaccine, according to new data from the Centers for Disease Control and Prevention (CDC).
As of Dec. 18, 3,150 people reported what the agency terms “Health Impact Events” after getting vaccinated.
The definition of the term is: “unable to perform normal daily activities, unable to work, required care from doctor or health care professional.”
As The Epoch Times’ Zachary Stieber reports, the people reporting the negative effects reported them through V-safe, a smartphone application. The tool uses text messages and web surveys to provide personalized health check-ins and allows users to quickly tell the CDC if they are experiencing side effects.
The CDC and Pfizer, which produces the vaccine with BioNTech, didn’t respond to request for comments.
The information was presented by Dr. Thomas Clark, a CDC epidemiologist, to the Advisory Committee on Immunization Practices, an independent panel that provides recommendations to the agency, on Saturday.
The CDC said that 272,001 doses of the vaccine were administered as of Dec. 19. That means most people who were vaccinated did not experience negative effects.
The CDC has identified six case reports of anaphylaxis, or severe allergic reaction, that occurred following vaccination with the new vaccine, Clark reported. Other case reports were reviewed and determined not to be of anaphylaxis.
In an update on Friday, the agency stressed that anyone who has ever had a severe allergic reaction to any ingredient in a COVID-19 vaccine should not get that vaccine. People with severe allergic reactions to other vaccines should consult their doctor about getting the new vaccine while those with a history of anaphylaxis not related to vaccines “may still get vaccinated.”
“CDC recommends that people with a history of severe allergic reactions not related to vaccines or injectable medications – such as allergies to food, pet, venom, environmental, or latex – may still get vaccinated,” the CDC said.
“People with a history of allergies to oral medications or a family history of severe allergic reactions, or who might have a milder allergy to vaccines (no anaphylaxis) – may also still get vaccinated.”
Anyone who experiences anaphylaxis after getting the first vaccine should not get the second shot, the CDC said. COVID-19 vaccines are meant to be given across two doses, spaced about three weeks apart.
At least five healthcare workers in Alaska experienced adverse reactions after getting the Pfizer vaccine, the Anchorage Daily News reported. One of two experiencing adverse reactions at the Bartlett Regional Hospital required treatment at the hospital for at least two nights.
An Illinois hospital halted vaccinations after four workers suffered adverse reactions.
Possible Adverse Effect: Death
Death could be an adverse side effect of those hyped-up vaccines.
The last half of the video offers evidence she died, despite the counter narrative.
And more casualties reported -
Tennessee Nurse Kalilah Mitchell got Bell’s Palsy from Covid Vax, warns others (1-min) - They do not care about us. Do not take this vaccination.
A nurse in California tested positive for COVID-19 more than a week after receiving Pfizer Inc’s vaccine. Matthew W., 45. Six days after taking it, Six days later on Christmas Eve, he became sick after working a shift in the COVID-19 unit. He got the chills and later came down with muscle aches and fatigue.
But this is all normal?
More incoming news about adverse effects - Death
After an Israeli man reportedly died just 2 hours after receiving his first dose of the COVID-19 vaccine, authorities in the Swiss Canton of Lucerne said on Wednesday that one of the first people in the country to receive the vaccine has died…
With the vaccine not providing immediate immunity to the coronavirus, over two hundred Israeli citizens have been diagnosed with the disease days after getting the Pfizer/BioNTech jabs.
Mexican doctor hospitalized after receiving COVID-19 vaccine. Mexican authorities said they are studying the case of a 32-year-old female doctor who was hospitalized after receiving the Pfizer-BioNTech COVID-19 vaccine.
Portugal - 41-year-old pediatric operational assistant dies two days after she received her first dose of the Covid vaccine. The woman was one of 538 health professionals at Porto’s IPO cancer hospital who received their first jabs of the BioNTech/ Pfizer vaccine last Wednesday.
Frontline workers with top-priority access to the COVID-19 vaccine are refusing to take it – good for them:
Deborah Tilli, 27-Year-Old Canadian Healthcare Worker, Faints and Suffers Multiple Seizures After Pfizer Experimental COVID Vaccine
Dr. Gregory Michael, a 56-year-old OBGYN at Mountain Sinai Medical Center (MSMC) in Miami Beach, Florida, died on Monday after sustaining a hemorrhagic stroke due to insufficient blood platelets.
Heidi Neckelmann (Dr. Michael’s wife):
The love of my life, my husband Gregory Michael MD an Obstetrician that had his office in Mount Sinai Medical Center in Miami Beach Died the day before yesterday due to a strong reaction to the COVID vaccine. He was a very healthy 56 year old, loved by everyone in the community delivered hundreds of healthy babies and worked tireless through the pandemic.
He was vaccinated with the Pfizer vaccine at MSMC on December 18, 3 days later he saw a strong set of petechiae on his feet and hands which made him seek attention at the emergency room at MSMC. The CBC that was done at his arrival showed his platelet count to be 0 (A normal platelet count ranges from 150,000 to 450,000 platelets per microliter of blood.) he was admitted in the ICU with a diagnosis of acute ITP caused by a reaction to the COVID vaccine. A team of expert doctors tried for 2 weeks to raise his platelet count to no avail. Experts from all over the country were involved in his care. No matter what they did, the platelets count refused to go up. He was conscious and energetic through the whole process but 2 days before a last resort surgery, he got a hemorrhagic stroke caused by the lack of platelets that took his life in a matter of minutes. He was a pro vaccine advocate that is why he got it himself. I believe that people should be aware that side effects can happened, that it is not good for everyone and in this case destroyed a beautiful life, a perfect family, and has affected so many people in the community
Do not let his death be in vain please save more lives by making this information news.
5850 doses of Covid-19 vaccine arrive in Gibraltar with vaccination programme starting Sunday
Saturday the 9th of January: an historic day for Gibraltar, bringing a ray of hope.
The first consignment of the Covid 19 vaccine arrived on the Rock at 17.55hrs.
It was stored in special freezers at St Bernard’s Hospital.
Early on Sunday morning, the first doses were set to be administered to front line staff and residents of Elderly Residential Services.
Priest who volunteered for COVID-19 vaccine trial dies.
John Fields was communications director of Ukrainian Catholic Archeparchy of Philadelphia. He had participated in the third and final phase of Moderna’s COVID-19 vaccine trial.
The Deceptive Spin - Just a few months ago, a person who died of a heart attack, was labeled as dying of Covid. Now, a person who dies after getting the Covid vaccine is labled as dying of a heart attack.
A risk analysis - this is an experimental vaccine which does not protect people from getting covid -
Brant Griner’s mom took the risk -